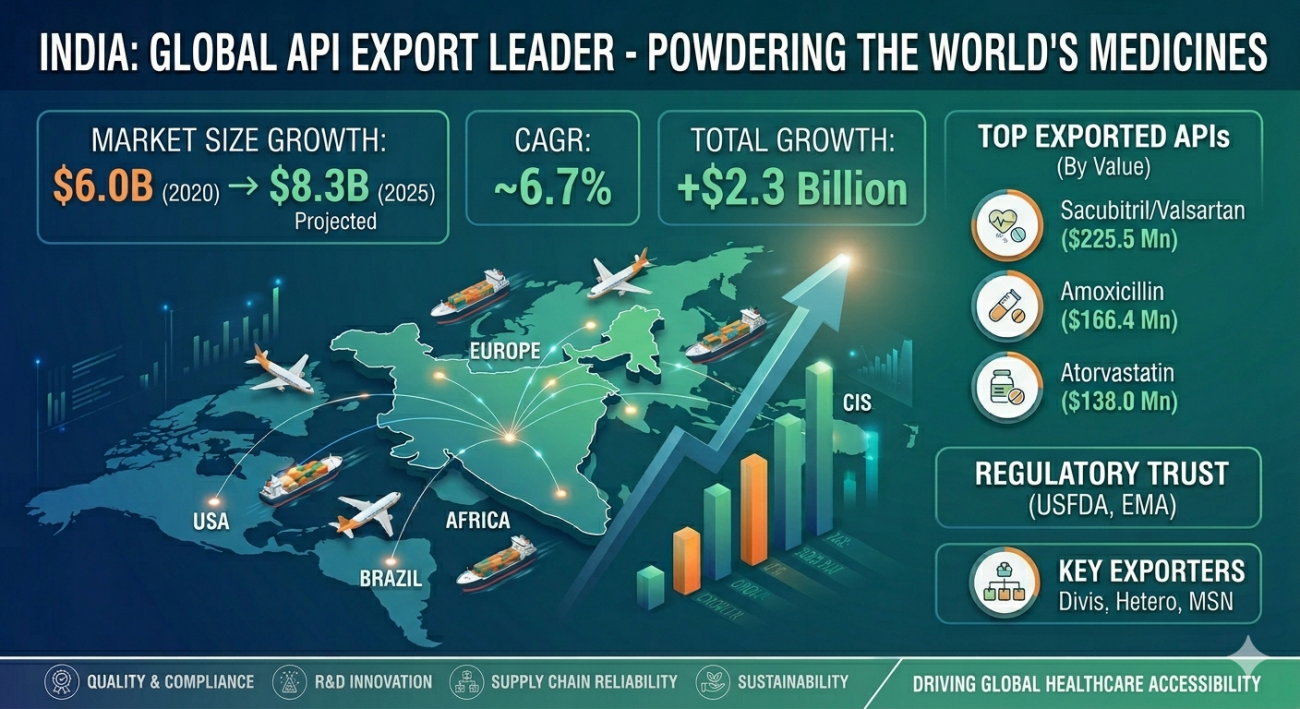
The India API export market has transformed from a domestic manufacturing base into the primary engine of the global pharmaceutical supply chain. Over the last five years, India has demonstrated unparalleled resilience, scaling its export value from $6.0 Billion in 2020 to a projected $8.3 Billion by 2025.
This comprehensive analysis explores how India became the ‘Pharmacy of the World’ through Active Pharmaceutical Ingredients (APIs). From basic generic drugs to highly complex biological molecules, the API export India ecosystem is evolving at an exponential rate.
1. India API Export Market Growth Analysis (2020–2025)
The India API export market has witnessed a golden era of growth. Historically, India was seen as a secondary supplier, but the disruptions of 2020 proved that India is the only nation capable of maintaining the global drug supply during a crisis. Between 2020 and 2025, the market expanded with a CAGR of 6.7%.
The Shift from Volume to Value
This growth is not just about quantity; it is about “Value Migration.” Indian manufacturers are moving away from low-margin products like Ibuprofen and Paracetamol towards high-margin specialty molecules. The total value addition of +$2.3 Billion in five years signifies a shift towards research-intensive manufacturing.
According to recent data from the Pharmaceuticals Export Promotion Council of India (PHARMEXCIL), the demand for Indian APIs in regulated markets like the US and EU has surged by 12% annually.
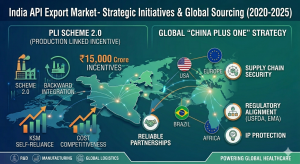
2. Strategic Impact of the PLI Scheme 2.0
One of the biggest turning points for the India API market size was the introduction of the Production Linked Incentive (PLI) scheme. The government recognized that over-dependence on imports for Key Starting Materials (KSMs) was a national security risk.
Reducing Import Dependency
With an allocation of nearly ₹15,000 crores, the PLI scheme has successfully incentivized the local production of 41 critical bulk drugs. This has lowered the landing cost of raw materials, making pharmaceutical API exports from India much more competitive in price-sensitive markets like Southeast Asia and Africa.
-
Self-Reliance: Reduction in KSM imports from China by 25%.
-
Capacity Building: Over 50 new manufacturing plants commissioned under PLI 1.0 and 2.0.
3. Geopolitics: India vs China (The China Plus One Strategy)
The global pharmaceutical industry is currently undergoing a massive structural shift known as “China Plus One.” International pharmaceutical giants are diversifying their supply chains to reduce reliance on China. The API export India sector has been the biggest beneficiary of this trend.
Why Global Firms are Choosing India:
-
Compliance Superiority: India has more USFDA-approved plants than any other country outside the US. You can track these approvals on the USFDA Official Database.
-
Intellectual Property: India’s legal system provides stronger IP protection, crucial for manufacturing patented molecules.
-
Communication: The ease of doing business in English makes India a preferred partner for Western MNCs.
4. Therapeutic Market Segmentation
The export market is divided into several high-growth therapeutic segments. Understanding these segments is crucial for investors and exporters alike.
Cardiovascular (CVS)
The largest segment by value, driven by APIs like Atorvastatin and Rosuvastatin. As lifestyle diseases increase globally, India’s role in supplying these life-saving molecules becomes paramount.
Oncology & High-Potency APIs (HPAPIs)
The fastest-growing niche. Manufacturing oncology APIs requires specialized containment facilities. Indian firms like Dr. Reddy’s and Natco Pharma are leading this transition toward complex chemotherapy agents.
Anti-Infectives
India continues to dominate the global supply of Penicillins and Cephalosporins. During the pandemic, Indian exporters ensured that antibiotics reached over 150 countries without delay.
5. Analysis of Top Exported API Molecules (Detailed Table)
| API Molecule | Estimated Export Value | Primary Market | Growth Rate (YoY) |
| Sacubitril/Valsartan | $225.5 Million | USA, Europe | 14% |
| Amoxicillin Trihydrate | $166.4 Million | Global (WHO) | 5% |
| Atorvastatin Calcium | $138.0 Million | USA, Brazil | 8% |
| Metformin HCl | $115.0 Million | Worldwide | 4% |
| Ritonavir | $95.0 Million | Africa, SE Asia | 11% |
6. Key Global Destinations & Trade Corridors
The India API export market footprint is truly global. While the United States remains the largest single market (contributing over $671.8 Mn in revenue), new corridors are opening up.
-
Brazil: A critical hub for generic production in Latin America.
-
Germany: The gateway to the European Union market.
-
Vietnam & Indonesia: Emerging markets where Indian APIs are replacing high-cost European alternatives.
7. Leading Players in the Indian API Sector
Innovation-led companies are the backbone of this success.
-
Divis Laboratories: World leader in custom synthesis and large-scale manufacturing.
-
MSN Laboratories: Known for the highest number of DMF (Drug Master File) filings.
-
Hetero Labs: The dominant force in anti-retroviral APIs for HIV treatment.
8. Regulatory Standards: USFDA, EMA & WHO-GMP
Quality is the currency of the API export India industry. Indian facilities are now moving towards ‘Paperless Audits’ and real-time monitoring to maintain zero-observation USFDA records.
The European Medicines Agency (EMA) has also streamlined its inspection process for Indian sites, recognizing the high standards of Good Manufacturing Practices (GMP) maintained in hubs like Hyderabad and Vizag.
9. Logistics, Supply Chain & Export Documentation
Exporting APIs is not just about chemistry; it’s about logistics.
-
Cold Chain Integrity: Many APIs require temperature-controlled shipping (2°C to 8°C).
-
Dangerous Goods (DG) Handling: Solvents and reactive intermediates require specialized certification.
-
Documentation: Key documents include the Certificate of Analysis (CoA), Drug Master File (DMF), and Certificate of Suitability (CEP).
10. The Strategic Shift Towards CDMO Services
The rise of Contract Development and Manufacturing Organizations (CDMOs) is increasing profit margins by 15-20%. Indian firms are no longer just “suppliers”; they are “partners” who help global pharma companies develop molecules from the clinical trial stage to commercial launch.
11. Green Chemistry and Environmental ESG
Environmental, Social, and Governance (ESG) norms are becoming mandatory for exporting to Europe.
-
Zero Liquid Discharge (ZLD): Indian plants are recycling 100% of their wastewater.
-
Green Catalysis: Using enzymes instead of toxic chemicals to synthesize APIs.
-
Carbon Footprint: Reducing the energy intensity of chemical reactions.
12. Industry 4.0: AI and Automation in Pharma
Artificial Intelligence (AI) is being used to:
-
Predict impurity profiles during the reaction.
-
Optimize the yield of expensive molecules.
-
Reduce batch failures in pharmaceutical API exports from India.
Automation is also helping in reducing human error in documentation, ensuring that every export batch meets global compliance standards.
13. Critical Challenges & Risk Mitigation
Despite the success, the industry faces hurdles:
-
Raw Material Volatility: Fluctuating prices of basic chemicals.
-
Regulatory Scrutiny: Increasing frequency of surprise inspections.
-
Global Competition: New manufacturing hubs emerging in Vietnam and Mexico.
Mitigation: Backward integration (making your own raw materials) is the primary strategy used by top Indian firms to stay ahead.
14. Future Outlook: The Road to 2030
By 2030, the India API export market is expected to reach $12 Billion. The focus will shift from small molecules to:
-
Biologics and Biosimilars: Large molecule drugs.
-
mRNA Technology: Building on the vaccine success of 2021.
-
Peptides: High-value niche molecules for metabolic disorders.
Frequently Asked Questions (FAQs)
Q1: What is the India API export market size in 2025?
The market is projected to reach $8.3 Billion by 2025, growing at a CAGR of 6.7%.
Q2: How has the PLI scheme helped exporters?
The PLI scheme provides financial incentives for local manufacturing, reducing production costs by 10-15% and decreasing reliance on Chinese raw materials.
Q3: Which countries are the top buyers of Indian APIs?
The USA is the largest buyer, followed by Brazil, Germany, UK, and South Africa.
Q4: What is a DMF in API export?
A Drug Master File (DMF) is a confidential document submitted to regulatory agencies (like USFDA) that provides detailed information about the manufacturing process of an API.
Sources: